Investigational New Drug (IND) Application Toolkit for Gene Therapy
The Somatic Cell Genome Editing (SCGE) Consortium is an NIH Common Fund program created to accelerate translational research in genome editing, and a primary goal of this initiative is to submit IND applications for novel gene editing therapies. As part of this work, we have created a collection of useful resources, templates, and tools for filling out an IND application.
Common Technical Document (CTD)
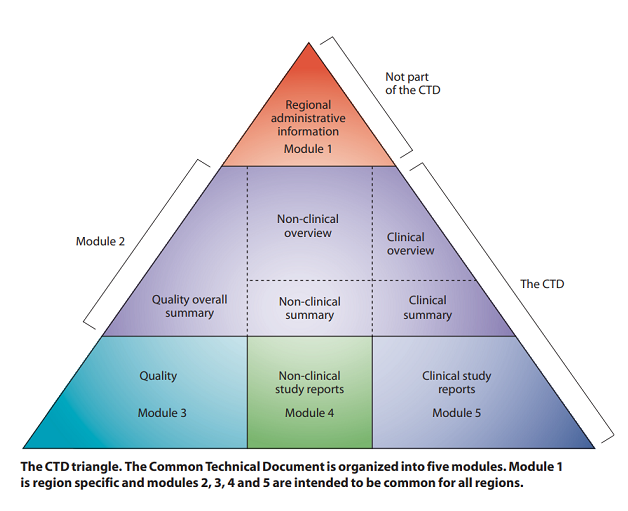
The eCTD contains an electronic table of contents also referred to as a backbone that manages all the metadata for an application. This backbone is broken down into five modules. Documents are placed appropriately into modules, which are graphically presented as the CTD Triangle.
Module 1 references regional information such as forms, cover letter, labeling, and investigational brochures.
Module 2 references summaries such as quality, clinical, and non-clinical summaries.
Module 3 references quality information.
Module 4 references non-clinical information.
Module 5 references clinical information.
FDA Submission Elements
| Section | Section Name | Required for submission | Submission Timing | Guidance | Regulation | Templates | Examples | Other Links | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Initial IND | Marketing | |||||||||||
| Module1 | ||||||||||||
| 1.1 | Forms | Yes | ||||||||||
| Form FDA 1571: Investigational New Drug Application (IND) | Yes | Include with every submission (initial IND AND all amendments) | ||||||||||
| Form FDA 356h: Application to Market a New Drug, Biologic, or an Antibiotic Drug for Human Use | No | Yes | Include with marketing applications only | |||||||||
| Form FDA 3397: User Fee Cover Sheet | No | Yes | Include with marketing applications only | |||||||||
| Form FDA 2252: Transmittal of Annual Reports for Drugs and Biologics for Human Use | No | Yes | Include with marketing applications only | |||||||||
| Form FDA 2253: Transmittal of Advertisements and Promotional Labeling for Drugs and Biologics for Human Use | No | Yes | Include with marketing applications, CDER INDs only | |||||||||
| Form FDA 2567: Transmittal of Labels and Circulars | No | Yes | Include with marketing applications only | |||||||||
| Form FDA 3674: Certification of Compliance | Yes | Include with initial IND and IND amendments (if applicable) | ||||||||||
| Form FDA 3792: Biosimilar User Fee Cover Sheet (BsUFA) | No | Yes | Required for biosimilars or generic drug applications only | |||||||||
| Form FDA 3794: Generic Drug User Fee Cover Sheet (GDUFA) | No | Yes | Required for biosimilars or generic drug applications only | |||||||||
| Form 1572: Statement of Investigator | Maybe | Include with initial IND and IND amendments (if applicable) | ||||||||||
| 1.2 | Cover Letter | Yes | ||||||||||
| 1.3 | Administrative information | Yes | ||||||||||
| 1.3.1 | Contact/Sponsor/Applicant Information | Yes | Initial IND only | |||||||||
| 1.3.1.1 | Change of Address or Corporate Name | No | Include in IND amendment (if applicable) | |||||||||
| 1.3.1.2 | Change in Contact/Agent | No | Include in IND amendment (if applicable) | |||||||||
| 1.3.1.3 | Change in Sponsor | No | Include in IND amendment (if applicable) | |||||||||
| 1.3.1.4 | Transfer of Obligation | No | Include in IND amendment (if applicable) | |||||||||
| 1.3.1.5 | Change in Ownership of an Application | No | Yes | Include with marketing applications only | ||||||||
| 1.3.2 | Field Copy Certification | No | Yes | Include with marketing applications only | ||||||||
| 1.3.3 | Debarment Certification | No | Yes | Include with marketing applications only | ||||||||
| 1.3.4 | Financial certification and disclosure | No | Yes | Include with marketing applications only | ||||||||
| 1.3.5 | Patent and exclusivity | No | Yes | |||||||||
| 1.3.5.1 | Patent Information | No | Yes | Include with marketing applications only | ||||||||
| 1.3.5.2 | Patent Certification | No | Yes | Include with marketing applications only | ||||||||
| 1.3.5.3 | Exclusivity request | No | Yes | Include with marketing applications only | ||||||||
| 1.3.6 | Tropical disease priority review voucher | No | Not applicable to gene therapies | |||||||||
| 1.4 | References | Maybe | ||||||||||
| 1.4.1 | Letter of authorization | Maybe | Include in initial IND or amendments (if applicable) | |||||||||
| 1.4.2 | Statement of right of reference | Maybe | Include in initial IND or amendments (if applicable) | |||||||||
| 1.4.3 | List of authorized persons to incorporate by reference | Maybe | Include in initial IND or amendments (if applicable) | |||||||||
| 1.4.4 | Cross reference to previously submitted information | Maybe | Include in initial IND or amendments (if applicable) | |||||||||
| 1.5 | Application Status | No | ||||||||||
| 1.5.1 | Withdrawal of an IND | No | Include in IND amendment (if applicable) | |||||||||
| 1.5.2 | Inactivation request | No | Include in IND amendment (if applicable) | |||||||||
| 1.5.3 | Reactivation request | No | Include in IND amendment (if applicable) | |||||||||
| 1.5.4 | Reinstatement request | No | Include in IND amendment (if applicable) | |||||||||
| 1.5.5 | Withdrawal of an unapproved BLA, NDA, ANDA, or Supplement | No | Yes | Include with marketing applications only | ||||||||
| 1.5.6 | Withdrawal of listed drug | No | Yes | Include with marketing applications only | ||||||||
| 1.5.7 | Withdrawal of approval of an application or revocation of license | No | Yes | Include with marketing applications only | ||||||||
| 1.6 | Meetings | Maybe | ||||||||||
| 1.6.1 | Meeting request | Maybe | Initial IND or IND amendments (if applicable) | |||||||||
| 1.6.2 | Meeting background materials | Maybe | Initial IND or IND amendments (if applicable) | |||||||||
| 1.6.3 | Correspondence regarding meetings | Maybe | Initial IND or IND amendments (if applicable) | |||||||||
| 1.7 | Fast Track | Maybe | ||||||||||
| 1.7.1 | Fast track designation request | Maybe | Any submission prior to marketing application | |||||||||
| 1.7.2 | Fast track designation withdrawal request | No | IND amendment only (if applicable) | |||||||||
| 1.7.3 | Rolling review request | No | Yes | Include with marketing applications only | ||||||||
| 1.7.4 | Correspondence regarding fast track/rolling review | No | Yes | Include with marketing applications only | ||||||||
| 1.8 | Special protocol assessment request | No | ||||||||||
| 1.8.1 | Clinical study | No | Any submission prior to marketing application | |||||||||
| 1.8.2 | Carcinogenicity study | No | Any submission prior to marketing application | |||||||||
| 1.8.3 | Stability study | No | Any submission prior to marketing application | |||||||||
| 1.8.4 | Animal efficacy study for approval under the animal rule | No | Any submission prior to marketing application | |||||||||
| 1.9 | Pediatric administrative information | No | ||||||||||
| 1.9.1 | Request for waiver of pediatric studies | No | Any submission prior to marketing application | |||||||||
| 1.9.2 | Request for deferral of pediatric studies | No | Any submission prior to marketing application | |||||||||
| 1.9.3 | Request for pediatric exclusivity determination | No | Any submission prior to marketing application | |||||||||
| 1.9.4 | Proposed pediatric study request and amendments | No | Any submission prior to marketing application | |||||||||
| 1.9.6 | Other correspondence regarding pediatric exclusivity or study plans | No | Any submission prior to marketing application | |||||||||
| 1.10 | Dispute Resolution | No | ||||||||||
| 1.10.1 | Request for dispute resolution | No | Initial IND or IND amendments (if applicable) | |||||||||
| 1.10.2 | Correspondence related to dispute resolution | Maybe | Initial IND or IND amendments (if applicable) | |||||||||
| 1.11 | Information Amendment: Information not Covered under Modules 2 to 5 | No | ||||||||||
| 1.11.1 | Quality information amendment | No | IND amendment only (if applicable) | |||||||||
| 1.11.2 | Nonclinical/Safety information amendment | No | IND amendment only (if applicable) | |||||||||
| 1.11.3 | Clinical/Efficacy information amendment | No | IND amendment only (if applicable) | |||||||||
| 1.11.4 | Multiple module information amendment | No | IND amendment only (if applicable) | |||||||||
| 1.12 | Other correspondence | Maybe | ||||||||||
| 1.12.1 | Pre IND correspondence | Maybe | Initial IND (if applicable) | |||||||||
| 1.12.2 | Request to charge for clinical trial | Maybe | Initial IND or IND amendments (if applicable) | |||||||||
| 1.12.3 | Request to charge for expanded access | Maybe | Initial IND or IND amendments (if applicable) | |||||||||
| 1.12.4 | Request for comments and advice | Maybe | Initial IND or IND amendments (if applicable) | |||||||||
| 1.12.5 | Request for a waiver | No | Initial IND or IND amendments (if applicable) | |||||||||
| 1.12.6 | Exception from informed consent for emergency research | No | Not applicable to gene therapies | |||||||||
| 1.12.7 | Public disclosure statement for exception from informed consent for emergency research | No | Not applicable to gene therapies | |||||||||
| 1.12.8 | Correspondence regarding exception from informed consent for research | No | Not applicable to gene therapies | |||||||||
| 1.12.9 | Notification of discontinuation of clinical trial | No | IND amendment only (if applicable) | |||||||||
| 1.12.10 | Generic drug enforcement act statement | No | Not applicable to gene therapies | |||||||||
| 1.12.11 | ANDA basis for submission statement | No | Not applicable to gene therapies | |||||||||
| 1.12.12 | Comparison of generic drug and reference listed drug | No | Not applicable to gene therapies | |||||||||
| 1.12.13 | Request for waiver for in vivo studies | No | Not applicable to gene therapies | |||||||||
| 1.12.14 | Environmental analysis | Yes | Required in initial IND and marketing application | |||||||||
| 1.12.15 | Request for waiver of in vivo bioavailability studies | No | Not applicable to gene therapies | |||||||||
| 1.12.16 | Field alert reports | No | IND amendment only (if applicable) | |||||||||
| 1.12.17 | Orphan drug designation | Maybe | Initial IND or IND amendments (if applicable) | |||||||||
| 1.12.18 | Regenerative medicine advanced therapy (RMAT) designation | Maybe | IND amendments (if applicable) | |||||||||
| 1.13 | Annual Report | No | ||||||||||
| 1.13.1 | Summary for nonclinical studies | No | IND amendments (annual reports only and if applicable) | |||||||||
| 1.13.2 | Summary of clinical pharmacology information | No | IND amendments (annual reports only and if applicable) | |||||||||
| 1.13.3 | Summary of safety information | No | IND amendments (annual reports only and if applicable) | |||||||||
| 1.13.4 | Summary of labeling changes | No | IND amendments (annual reports only and if applicable) | |||||||||
| 1.13.5 | Summary of manufacturing changes | No | IND amendments (annual reports only and if applicable) | |||||||||
| 1.13.6 | Summary of microbiological changes | No | IND amendments (annual reports only and if applicable) | |||||||||
| 1.13.7 | Summary of other significant new information | No | IND amendments (annual reports only and if applicable) | |||||||||
| 1.13.8 | Individual study information | No | IND amendments (annual reports only and if applicable) | |||||||||
| 1.13.9 | General investigational plan | No | IND amendments (annual reports only and if applicable) | |||||||||
| 1.13.10 | Foreign marketing | No | IND amendments (annual reports only and if applicable) | |||||||||
| 1.13.11 | Distribution data | No | IND amendments (annual reports only and if applicable) | |||||||||
| 1.13.12 | Status of postmarketing study commitments and requirements | No | Yes | Marketing application amendments (annual reports only and if applicable) | ||||||||
| 1.13.13 | Status of other postmarketing studies and requirements | No | Yes | Marketing application amendments (annual reports only and if applicable) | ||||||||
| 1.13.14 | Log of outstanding regulatory business | No | IND amendments (annual reports only and if applicable) | |||||||||
| 1.13.15 | Development safety update report (DSUR) | No | IND amendments (annual reports only) | |||||||||
| 1.14 | Labeling | Yes | ||||||||||
| 1.14.1 | Draft Labeling | No | Yes | |||||||||
| 1.14.1.1 | Draft carton and container labels | No | Yes | |||||||||
| 1.14.1.2 | Annotated draft labeling text | No | Yes | |||||||||
| 1.14.1.3 | Draft labeling text | No | Yes | |||||||||
| 1.14.1.4 | Label comprehension studies | No | Yes | Only if required by FDA | ||||||||
| 1.14.1.5 | Labeling History | No | Yes | |||||||||
| 1.14.2 | Final labeling | No | Yes |
|
||||||||
| 1.14.2.1 | Final carton or container labels | No | Yes | Marketing applications only | ||||||||
| 1.14.2.2 | Final package insert (package inserts, patient information, Medication guides) | No | Yes | Marketing applications only | ||||||||
| 1.14.2.3 | Final labeling text | No | Yes | Marketing applications only | ||||||||
| 1.14.3 | Listed Drug Labeling | No | Yes | |||||||||
| 1.14.3.1 | Annotated comparison with listed drug | No | Yes | Included with generic drug applications | ||||||||
| 1.14.3.2 | Approved labeling text for listed drug | No | Yes | Included with generic drug applications | ||||||||
| 1.14.3.3 | Labeling text for reference listed drug | No | Yes | Included with generic drug applications | ||||||||
| 1.14.4 | Investigational drug labeling | Yes | ||||||||||
| 1.14.4.1 | Investigational brochure | Maybe | Initial IND or IND amendments (if applicable) | |||||||||
| 1.14.4.2 | Investigational drug labeling | Yes | Required in initial IND application or in amendments if changes are made | |||||||||
| 1.14.5 | Foreign labeling | No | Marketing applications (if applicable) | |||||||||
| 1.14.6 | Product labeling for 2253 submissions | No | Include with marketing applications, CDER INDs only | |||||||||
| 1.15 | Promotional material [promotional-material-audience-type] | No | ||||||||||
| 1.15.1 | Correspondence relating to promotional materials | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.15.1.1 | Request for advisory comments on launch materials | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.15.1.2 | Request for advisory comments on non-launch materials | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.15.1.3 | Presubmission of launch promotional materials for accelerated approval products | No | Yes | Include with marketing applications that are seeking accelerated approval designation (prior to launch) | ||||||||
| 1.15.1.4 | Presubmission of non-launch promotional materials for accelerated approval products | No | Yes | Include with marketing applications that are seeking accelerated approval designation (after launch phase) | ||||||||
| 1.15.1.5 | Pre-dissemination review of television ads | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.15.1.6 | Response to untitled letter or warning letter | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.15.1.7 | Response to information request | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.15.1.8 | Correspondence accompanying materials previously missing or rejected | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.15.1.9 | Withdrawal request | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.15.1.10 | Submission of annotated references | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.15.1.11 | General correspondence | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.15.2 | Materials attribute = [promotional-material-doc-type] | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.15.2.1 | Material = [promotional-material-type, material-id, issue-date] | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.15.2.1.1 | Clean version | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.15.2.1.2 | Annotated version | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.15.2.1.3 | Annotated labeling version | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.15.2.1.4 | Annotated references | No | Yes | Include in marketing application (if applicable) | ||||||||
| 1.16 | Risk management plan | No | ||||||||||
| 1.16.1 | Risk Management (non-REMS) | No | Not required, but FDA highly encourages sponsors to proactively manage known or anticipated risks | |||||||||
| 1.16.2 | Risk Evaluation and Mitigation Strategy (REMS) | No | ||||||||||
| 1.16.2.1 | Final REMS | No | Yes | Include in marketing application, postmarketing amendment | ||||||||
| 1.16.2.2 | Draft REMS | No | Yes | Include in marketing application, postmarketing amendment | ||||||||
| 1.16.2.3 | REMS Assessment | No | Yes | Include in marketing application, postmarketing amendment | ||||||||
| 1.16.2.4 | REMS Assessment Methodology | No | Yes | Include in marketing application, postmarketing amendment | ||||||||
| 1.16.2.5 | REMS Correspondence | No | Yes | Include in marketing application, postmarketing amendment | ||||||||
| 1.16.2.6 | REMS Modification History | No | Yes | Include in marketing application, postmarketing amendment | ||||||||
| 1.17 | Postmarketing studies | No | ||||||||||
| 1.17.1 | Correspondence regarding postmarketing commitments | No | Yes | Marketing applications only | ||||||||
| 1.17.2 | Correspondence regarding postmarketing requirements | No | Yes | Marketing applications only | ||||||||
| 1.18 | Proprietary names | No | ||||||||||
| 1.18.1 | Proprietary names | No | At any point up to and including marketing application | |||||||||
| 1.18.2 | Biological Proper Name Suffix | No | At any point up to and including marketing application | |||||||||
| 1.19 | Pre-EUA and EUA | No | ||||||||||
| 1.20 | General investigational plan for initial IND | Yes | ||||||||||
| Section | Section Name | Required for submission | Submission Timing | Guidance | Regulation | Templates | Examples | Other Links | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Initial IND | Marketing | |||||||||||
| Module2 | ||||||||||||
| 2.2 | Introduction to Summary | Yes | ||||||||||
| 2.3 | Quality Overall Summary | Yes | ||||||||||
| 2.3.I | Introduction | Yes | Initial IND application, revise as necessary | |||||||||
| 2.3.S | Drug Substance [substance (O), manufacturer (O)] | Yes | ||||||||||
| 2.3.P | Drug Product [product (O), dosage form (O)] | Yes | ||||||||||
| 2.3.A | Appendices | |||||||||||
| 2.3.A.1 | Facilities and Equipment [facility (O)] | Yes | Initial IND application, revise as necessary | |||||||||
| 2.3.A.2 | Adventitious Agents Safety Evaluation [component (O)] | Yes | Initial IND application, revise as necessary | |||||||||
| 2.3.A.3 | Excipients | Maybe | Initial IND application, revise as necessary | |||||||||
| 2.3.R | Regional Information | Maybe | Initial IND application, revise as necessary | |||||||||
| 2.4 | Nonclinical Overview | Yes | ||||||||||
| 2.5 | Clinical Overview | Maybe | ||||||||||
| 2.6 | Nonclinical Written and Tabulated Summaries | |||||||||||
| 2.6.1 | Introduction | Yes | Initial IND application, revise as necessary | |||||||||
| 2.6.2 | Pharmacology Written Summary | Yes | Initial IND application, revise as necessary | |||||||||
| 2.6.3 | Pharmacology Tabulated Summary | Yes | Initial IND application, revise as necessary | |||||||||
| 2.6.4 | Pharmacokinetic Written Summary | Maybe | Initial IND application, revise as necessary | |||||||||
| 2.6.5 | Pharmacokinetic Tabulated Summary | Maybe | Initial IND application, revise as necessary | |||||||||
| 2.6.6 | Toxicology Written Summary | Yes | Initial IND application, revise as necessary | |||||||||
| 2.6.7 | Toxicology Tabulated Summary | Yes | Initial IND application, revise as necessary | |||||||||
| 2.7 | Clinical Summary | |||||||||||
| 2.7.1 | Summary of Biopharmaceutic Studies and Associated Analytical Methods | Maybe | As early as initial IND application (see note), completed by marketing application | |||||||||
| 2.7.2 | Summary of Clinical Pharmacology Studies | Maybe | As early as initial IND application (see note), completed by marketing application | |||||||||
| 2.7.3 | Summary of Clinical Efficacy [indication] | Maybe | As early as initial IND application (see note), completed by marketing application | |||||||||
| 2.7.4 | Summary of Clinical Safety | Maybe | As early as initial IND application (see note), completed by marketing application | |||||||||
| 2.7.5 | References - List of References | Maybe | As early as initial IND application (see note), completed by marketing application | |||||||||
| 2.7.6 | Synopses of Individual Studies | Maybe | As early as initial IND application (see note), completed by marketing application | |||||||||
| Section | Section Name | Required for submission | Submission Timing | Guidance | Regulation | Templates | Examples | Other Links | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Initial IND | Marketing | |||||||||||
| Module3 | ||||||||||||
| 3.2 | Body of Data |
|
||||||||||
| 3.2.S | Drug Substance [name, manufacturer] | |||||||||||
| 3.2.S.1 | General Information | Yes | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.2 | Manufacture | Yes | ||||||||||
| 3.2.S.2.1 | Manufacturer(s) | Yes | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.2.2 | Description of Manufacturing Process and Process Controls | Yes | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.2.3 | Control of Materials | Yes | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.2.4 | Controls of Critical Steps and Intermediates | Yes | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.2.5 | Process Validation and/or Evaluation | Maybe | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.2.6 | Manufacturing Process Development | Yes | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.3 | Characterization | |||||||||||
| 3.2.S.3.1 | Elucidation of Structure and Other Characteristics | Yes | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.3.2 | Impurities | Yes | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.4 | Control of Drug Substance | |||||||||||
| 3.2.S.4.1 | Specification | Yes | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.4.2 | Analytical Procedures | Yes | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.4.3 | Validation of Analytical Procedures | Maybe | Include in initial IND if requested, update in annual reports | |||||||||
| 3.2.S.4.4 | Batch Analyses | Yes | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.4.5 | Justification of Specification | Yes | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.5 | Reference Standards or Materials | Yes | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.6 | Container Closure Systems | Yes | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.7 | Stability | |||||||||||
| 3.2.S.7.1 | Stability Summary and Conclusions | Yes | Initial IND or IND amendments (if applicable) | |||||||||
| 3.2.S.7.2 | Post Approval Stability Protocol and Stability Commitment | No | Y | Prior to or with marketing application | ||||||||
| 3.2.S.7.3 | Stability Data | Maybe | Include initial data if applicable, update in annual reports | |||||||||
| 3.2.P | Drug Product [name, dosage form, manufacturer] | |||||||||||
| 3.2.P.1 | Description and Composition of the Drug Product | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.2 | Pharmaceutical Development | |||||||||||
| 3.2.P.3 | Manufacture | |||||||||||
| 3.2.P.3.1 | Manufacturer(s) | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.3.2 | Batch Formula | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.3.3 | Description of Manufacturing Process and Process Controls | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.3.4 | Controls of Critical Steps and Intermediates | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.3.5 | Process Validation and/or Evaluation | No | Required prior to initiation of Phase 3 manufacturing | |||||||||
| 3.2.P.4 | Control of Excipients | |||||||||||
| 3.2.P.4.1 | Specification(s) | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.4.2 | Analytical Procedures | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.4.3 | Validation of Analytical Procedures | Maybe | Include in initial IND if requested, update in annual reports | |||||||||
| 3.2.P.4.4 | Justification of Specifications | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.4.5 | Excipients of Human or Animal Origin | Maybe | Initial IND, update as needed | |||||||||
| 3.2.P.4.6 | Novel Excipients | Maybe | Initial IND, update as needed | |||||||||
| 3.2.P.5 | Control of Drug Product | |||||||||||
| 3.2.P.5.1 | Specification(s) | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.5.2 | Analytical Procedures | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.5.3 | Validation of Analytical Procedures | Yes | Include in initial IND if requested, update in annual reports | |||||||||
| 3.2.P.5.4 | Batch Analyses | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.5.5 | Characterization of Impurities | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.5.6 | Justification of Specification(s) | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.6 | Reference Standards or Materials | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.7 | Container Closure System | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.8 | Stability | |||||||||||
| 3.2.P.8.1 | Stability Summary and Conclusion | Yes | Initial IND, update as needed | |||||||||
| 3.2.P.8.2 | Post-approval Stability Protocol and Stability Commitment | No | Y | Prior to or with marketing application | ||||||||
| 3.2.P.8.3 | Stability Data | Yes | Initial IND, update as needed | |||||||||
| 3.2.A | Appendices | |||||||||||
| 3.2.A.1 | Facilities and Equipment [name, manufacturer] | Yes | Initial IND, update as needed | |||||||||
| 3.2.A.2 | Adventitious Agents Safety Evaluation [name, dosage form, manufacturer] | Yes | Initial IND, update as needed | |||||||||
| 3.2.A.3 | Novel Excipients | Maybe | Initial IND, update as needed | |||||||||
| 3.2.R | Regional Information | |||||||||||
| 3.3 | Literature References | |||||||||||
| Section | Section Name | Required for submission | Submission Timing | Guidance | Regulation | Templates | Examples | Other Links | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Initial IND | Marketing | |||||||||||
| Module4 | ||||||||||||
| 4.2 | Study Reports [study id_study Title] |
|
||||||||||
| 4.2.1 | Pharmacology Studies | Yes | ||||||||||
| 4.2.1.1 | Primary Pharmacodynamics | Yes | As early as initial IND application, revise as necessary | |||||||||
| 4.2.1.2 | Secondary Pharmacodynamics | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.1.3 | Safety Pharmacology | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.1.4 | Pharmacodynamic Drug Interactions | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.2 | Pharmacokinetic Studies | Yes | ||||||||||
| 4.2.2.1 | Analytical Methods and Validation Reports | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.2.2 | Absorption | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.2.3 | Distribution | Yes | As early as initial IND application, revise as necessary | |||||||||
| 4.2.2.4 | Metabolism | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.2.5 | Excretion | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.2.6 | Pharmacokinetic Drug Interactions | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.2.7 | Other Pharmacokinetic Studies | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3 | Toxicology Studies | Yes | ||||||||||
| 4.2.3.1 | Single-Dose Toxicity (in order by species, by route) | Yes | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.2 | Repeat-Dose Toxicity (species, route, duration, including supportive toxicokinetics) | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.3 | Genotoxicity | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.3.1 | In vitro | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.3.2 | In vivo (including supportive toxicokinetics evaluations) | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.4 | Carcinogenicity | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.4.1 | Long-term studies | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.4.2 | Short-or medium-term studies | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.4.3 | Other Studies | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.5 | Reproductive and Developmental Toxicity | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.5.1 | Fertility and early embryonic development | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.5.2 | Embryofetal development | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.5.3 | Prenatal and postnatal development, including maternal function | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.5.4 | Studies in which the offspring are dosed and/or further evaluated | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.6 | Local Tolerance | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.7 | Other Toxicity Studies | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.7.1 | Antigenicity | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.7.2 | Immunotoxicity | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.7.3 | Mechanistic studies | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.7.4 | Dependence | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.7.5 | Metabolites | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.7.6 | Impurities | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.2.3.7.7 | Other | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 4.3 | Literature References | |||||||||||
| Section | Section Name | Required for submission | Submission Timing | Guidance | Regulation | Templates | Examples | Other Links | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Initial IND | Marketing | |||||||||||
| Module5 | ||||||||||||
| 5.2 | Tabular Listing of All Clinical Studies | Maybe | ||||||||||
| 5.3 | Clinical Study Reports and Related Information [study id_study Title] | |||||||||||
| 5.3.1 | Reports of Biopharmaceutic Studies | |||||||||||
| 5.3.1.1 | Bioavailability (BA) Study Reports | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.3.1.2 | Comparative BA and Bioequivalence (BE) Study Reports | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.3.1.3 | In Vitro-In Vivo Correlation Study Reports | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.3.1.4 | Reports of Bioanalytical and Analytical Methods for Human Studies | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.3.2 | Reports of Studies Pertinent to Pharmocokinetics using Human Biomaterials | |||||||||||
| 5.3.2.1 | Plasma Protein Binding Study Reports | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.3.2.2 | Reports of Hepatic Metabolism and Drug Interaction Studies | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.3.2.3 | Reports of Studies Using Other Human Biomaterials | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.3.3 | Reports of Human Pharmacokinetic (PK) Studies | |||||||||||
| 5.3.3.1 | Healthy Subject PK and Initial Tolerability Study Reports | No | Not applicable to gene therapies, not tested in healthy subjects | |||||||||
| 5.3.3.2 | Patient PK and Initial Tolerability Study Reports | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.3.3.3 | Intrinsic Factor PK Study Reports | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.3.3.4 | Extrinsic Factor PK Study Reports | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.3.3.5 | Population PK Study Reports | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.3.4 | Reports of Human Pharmacodynamic (PD) Studies | |||||||||||
| 5.3.4.1 | Healthy Subject PD and PK/PD Study Reports | No | Not applicable to gene therapies, not tested in healthy subjects | |||||||||
| 5.3.4.2 | Patient PD and PK/PD Study Reports | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.3.5 | Reports of Efficacy and Safety Studies | |||||||||||
| 5.3.5.1 | Study Reports and Related Information of Controlled Clinical Studies Pertinent to the Claimed Indication | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.3.5.2 | Study Reports and Related Information of Uncontrolled Clinical Studies | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.3.5.3 | Reports of Analyses of Data From More Than One Study | |||||||||||
| 5.3.5.4 | Other Clinical Study Reports | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.3.6 | Reports of Postmarketing Experience | No | As early as initial IND application, revise as necessary | |||||||||
| 5.3.7 | Case Report Forms and Individual Patient Listings | Maybe | As early as initial IND application, revise as necessary | |||||||||
| 5.4 | Literature References | Yes | ||||||||||