About SCGE Toolkit
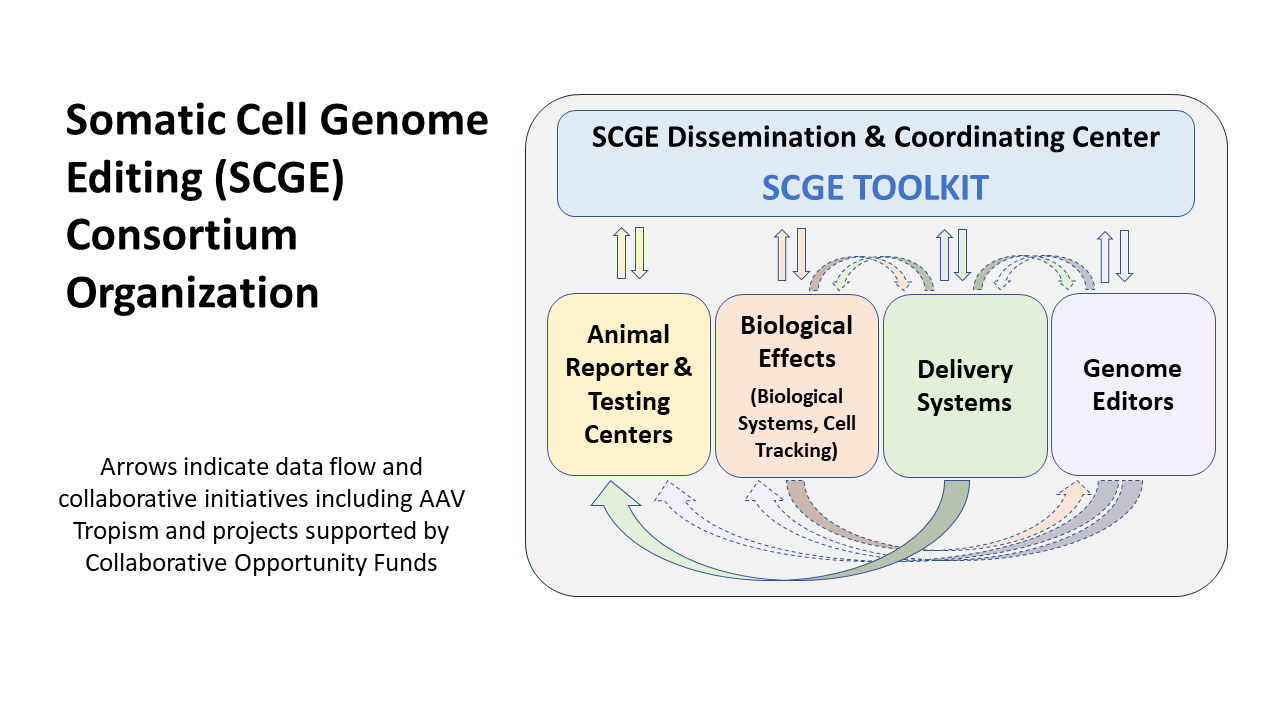
The Somatic Cell Genome Editing (SCGE) Consortium is supported by the NIH Common Fund through cooperative agreements by multiple National Institutes of Health (NIH) institutes to advance genome editing technologies to the clinic. In addition to scientific initiatives (below), the SCGE Dissemination and Coordinating Center (DCC) was funded (U24HG010423) to facilitate consortium interactions and scientific exchange and to collect and disseminate consortium generated data through the SCGE Toolkit to the broader biomedical research community.
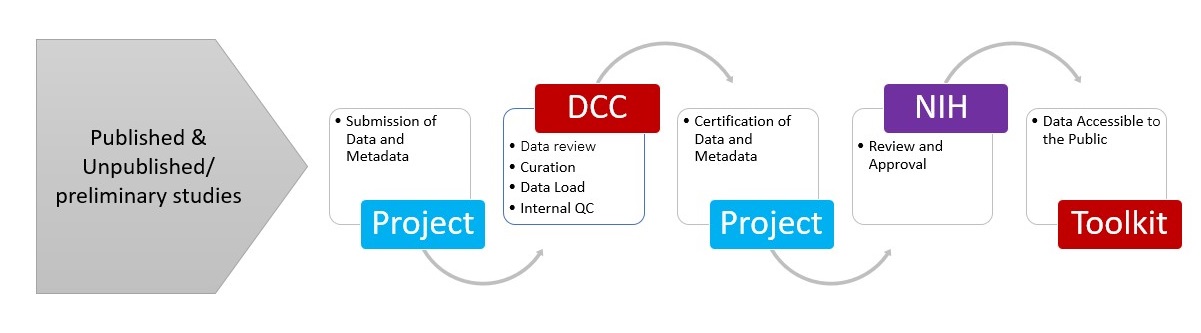
The Somatic Cell Genome Editing (SCGE) Toolkit team collects data and metadata from funded consortium laboratories, reviews, curates and standardizes data for loading into the Toolkit, works with the submitting laboratory and NIH program staff to certify data loading, and then makes data publicly accessible.
Toolkit Data Processing Pipeline
Somatic Cell Genome Editing (SCGE) Program Initiatives

Animal Reporting and
Testing Centers

Genome Editor
Projects

Delivery Systems
Initiative
Biological Effects
In Vivo Cell Tracking ProjectsThe objective of the program is to support the development of tools and technologies that will enable longitudinal monitoring and tracking of genome edited cells in humans to better assess the safety and efficacy of genome editing therapies. This will be accomplished through the development of innovative non-invasive technologies to label and track genome-edited cells in vivo, ideally in a clinically-relevant matter that has the potential to assess long-term safety in genome editing clinical trial participants.More...
Somatic Cell Genome Editing (SCGE) Toolkit Team
Principal Investigator
Co-Investigators
Anne Kwitek, PhD
Team Members
Mike Grzybowski, MS
Angela Lemke, BS
Jennifer Smith, MS
Jyothi Thota, MS
Marek Tutaj, MS
Monika Tutaj, PhD
Stacy Zacher, MS
Kent Brodie, MS
Former Members
Matt Hoffman, BS
Harika Nalabolu, MS